What Is THC (Tetrahydrocannabinol)?

What is THC?
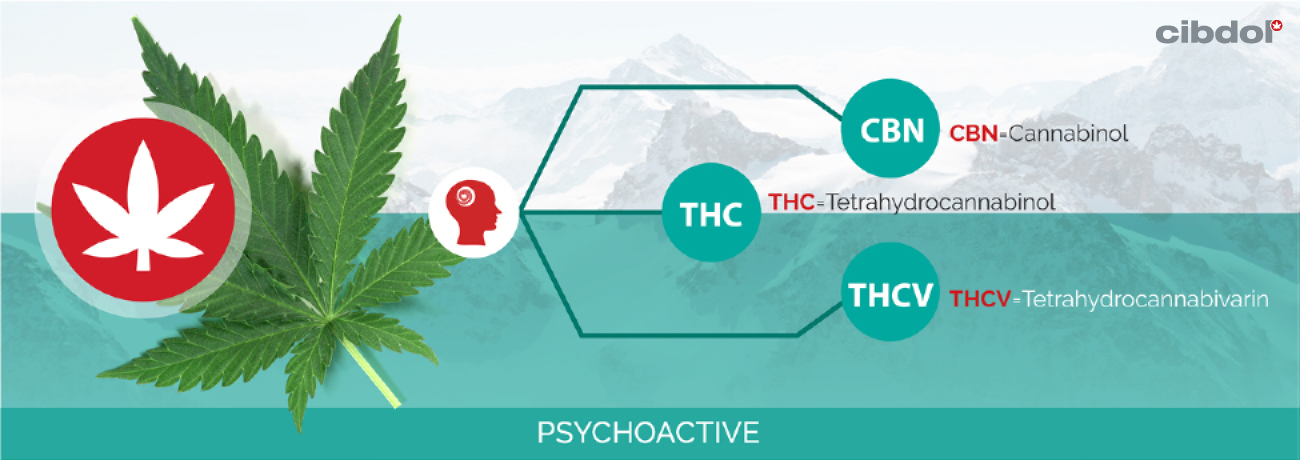
Tetrahydrocannabinol (THC) belongs to a class of organic chemical compounds called phytocannabinoids. Found within the Cannabis sativa species, THC is largely known for its psychoactive side effects. When consumed (smoked, vaporised, or eaten), THC primarily binds with CB1 receptors in the amygdala, cerebellum, and hippocampus.
Summary
Varieties of Cannabis sativa rich in THC (marijuana) are predominantly used recreationally. Cannabis is considered the most used illicit drug in the world.
• THC can be produced synthetically
• Potential short-term impairment of memory and coordination
• Potential increase in appetite, an effect commonly known as “the munchies”
• Responsible for inducing an altered mental state (high)
• Strong affinity for CB1 receptors linked to the endocannabinoid system
• First isolated in 1964
• In controlled doses, is linked as a possible treatment for several medical conditions
Supporting research
Due to THC’s Schedule I classification, research into its therapeutic application is limited. Studies can be conducted provided an administrative letter of authorisation is received first. Below you will find a selection of preclinical studies examining the effects of THC on nausea, multiple sclerosis, and pain relief.
Synthetic THC drug Dronabinol is approved by the US Food and Drug Administration (FDA) for the treatment of nausea and vomiting (CINV). A systematic review conducted in 2001 found that “cannabinoids have been used effectively for treating CINV since 1985”[1][2].
A systematic 2018 review of Cannabinoids for Treatment of M.S. symptoms found that there is “evidence to indicate benefits of treatment of spasticity and neuropathic pain in multiple sclerosis”. The researchers did highlight that cannabis use does include the risk of adverse side effects[3].
The Journal of Pain and Symptom Management published a study into the effect of THC:CBD extract and THC extract in patients with “intractable cancer-related pain”. Patients were randomised, and results compared against a placebo group. Overall, the THC:CBD extract group saw a significant reduction in pain measurement (30%)[4].
Legal status
In 1961, cannabis (because of its active constituent THC) was classified as a Schedule I narcotic. At the time, it was believed that there was “no currently accepted medical use and a high potential for abuse”.
The presence of THC in CBD oils and other cannabinoid products is considered legal as long as concentrations are below 0.2/0.3% (country-dependent).
A growing number of countries and American states are starting to amend legislation in favour of THC. In October 2018, Canada legalised cannabis for comprehensive recreational and medicinal use.
[1] World Health Organization. (2018, 7 juni). Cannabidiol (CBD) Critical Review Report. www.who.int. Geraadpleegd op 15 november 2021, van https://www.openaccessgovernment.org/who-perspective-on-cannabidiol/80838 [Source]
[2] Tramer, M. R. (2001). Cannabinoids for control of chemotherapy induced nausea and vomiting: quantitative systematic. BMJ, 323(7303), 16. https://doi.org/10.1136/bmj.323.7303.16 [Source]
[3] Rice, J., & Cameron, M. (2018). Cannabinoids for Treatment of MS Symptoms: State of the Evidence. Current Neurology and Neuroscience Reports, 18(8). https://doi.org/10.1007/s11910-018-0859-x [Source]
[4] Johnson, J. R., Burnell-Nugent, M., Lossignol, D., Ganae-Motan, E. D., Potts, R., & Fallon, M. T. (2010). Multicenter, Double-Blind, Randomized, Placebo-Controlled, Parallel-Group Study of the Efficacy, Safety, and Tolerability of THC:CBD Extract and THC Extract in Patients with Intractable Cancer-Related Pain. Journal of Pain and Symptom Management, 39(2), 167–179. https://doi.org/10.1016/j.jpainsymman.2009.06.008 [Source]
[1] World Health Organization. (2018, 7 juni). Cannabidiol (CBD) Critical Review Report. www.who.int. Geraadpleegd op 15 november 2021, van https://www.openaccessgovernment.org/who-perspective-on-cannabidiol/80838 [Source]
[2] Tramer, M. R. (2001). Cannabinoids for control of chemotherapy induced nausea and vomiting: quantitative systematic. BMJ, 323(7303), 16. https://doi.org/10.1136/bmj.323.7303.16 [Source]
[3] Rice, J., & Cameron, M. (2018). Cannabinoids for Treatment of MS Symptoms: State of the Evidence. Current Neurology and Neuroscience Reports, 18(8). https://doi.org/10.1007/s11910-018-0859-x [Source]
[4] Johnson, J. R., Burnell-Nugent, M., Lossignol, D., Ganae-Motan, E. D., Potts, R., & Fallon, M. T. (2010). Multicenter, Double-Blind, Randomized, Placebo-Controlled, Parallel-Group Study of the Efficacy, Safety, and Tolerability of THC:CBD Extract and THC Extract in Patients with Intractable Cancer-Related Pain. Journal of Pain and Symptom Management, 39(2), 167–179. https://doi.org/10.1016/j.jpainsymman.2009.06.008 [Source]





